Abstract
Introduction: asciminib is a first-in-class STAMP (Specifically Targeting the ABL Myristoyl Pocket) inhibitor that potently inhibits aberrant kinase activity of the BCR-ABL1 oncoprotein via allosteric binding. asciminib has shown high efficacy profile in heavily pretreated Chronic Myeloid Leukemia (CML) patients with an adequate safety profile in phase I and III clinical trials. However, data from the use of asciminib in real life setting are still scarce.
Methods: We gathered real-life retrospective data from 49 patients with BCR-ABL1 positive CML treated with asciminib (mean dose: 40 mg twice daily) between October 2018 and July 2021 at 33 institutions. The indication of asciminib was made according to the criterion of the attending physician and the drug was granted by Novartis under a controlled access program. Molecular biology tests were performed according to ELN guidelines and BCR-ABL/ABL ratios were expressed as % IS in all centers. Treatment responses were calculated with the patients at risk at each specific time points. For the event free survival (EFS), the events were treatment discontinuation due to any reason, progression or death. Data collection followed the local regulations for observational studies.
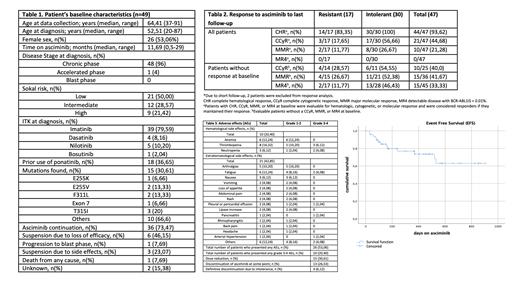
Results: Median time on asciminib was 11,69 months for the entire cohort. Patients' characteristics are displayed on Table 1. Most patients were heavily pretreated with at least 3 prior TKI lines in 45 patients (91,83%), 18 of them receiving prior Ponatinib. Switch to asciminib occurred due to intolerance in 32 patients and due to resistance in the remaining 17. Fifteen patients (30,61%) harbored mutations in BCR-ABL1 (3 with a T315 mutation). Regarding efficacy (Table 2), probability of reaching or maintaining previous responses were 94%, 45% and 21% for complete hematological response (CHR), complete cytogenetic response (CCyR) and major molecular response (MMR), respectively. Considering probabilities of improving previous response, rates were 40%, 42% and 33% for the same parameters. Probabilities to obtain CCyR and MMR in resistant and intolerant patients were 29% (4/14) vs 55% (6/11) and 27% (4/15) vs 52% (11/21), respectively. Amid the patients previously treated with Ponatinib, probabilities of reaching or maintaining previous response were 53% (9/17) and 35% (6/17) for CCyR and MMR respectively, and 30% (3/10), 23% (3/13) displayed improvement of response. Regarding responses in patients with mutations, 39% (5/13) achieved or maintained CCyR and 31% (4/13) MMR; whereas 20% (2/10) and 18% (2/11) improved such responses. Of the three patients with T315I mutation, one discontinued due to progression to advanced stages, and the rest maintained the previous response. With a median follow-up of 11,69 months, the estimated EFS was 80% (figure 1).
In terms of safety (Table 3), the most frequent extra-hematological adverse events (AE) were: fatigue (16,2%), joint pain (13,5%) and nausea (8,1%), most of them grade 1-2. Grade 3-4 AE were observed in 10% of patient (fatigue (2), cholestasis enzyme elevation (1), hypertension (1), pancreatitis (1) and pericardial effusion (1)). Thrombocytopenia was shown as the most frequent AE (16,3%), with 6% of patients suffering from grade 3-4. Dose reduction was required in 15 patients (30,6%). After a median follow up of 51 weeks, 73,5% of the patients remained on treatment. Only fourteen patients discontinued treatment due to progression or loss of efficacy, whereas 6% of patients discontinuing treatment due to intolerance.
Conclusions: The results presented are in line with the data obtained in clinical trials, positioning asciminib as a potential safe and efficacious treatment for CML patients with failure to several TKI lines.
Sanchez-Guijo: Novartis: Consultancy, Honoraria, Research Funding; Celgene/Bristol-Myers-Squibb,: Consultancy, Honoraria; Incyte: Consultancy, Honoraria; Pfizer: Consultancy, Honoraria; Takeda: Honoraria, Research Funding; Roche: Consultancy, Honoraria; Gilead: Consultancy, Honoraria; Amgen: Consultancy, Honoraria. Garcia Gutierrez: BMS: Consultancy, Honoraria, Research Funding; Novartis: Consultancy, Honoraria, Research Funding; Pfizer: Consultancy, Honoraria, Research Funding; Incyte: Consultancy, Honoraria, Research Funding.